The COVID-19 Vaccine: Questions and Answers

Back on January 5, 2020, the World Health Organization (WHO) published a summary of an unknown pneumonia outbreak in Wuhan, China. By March 11, 2020, this unknown pneumonia was known as COVID-19 and was declared a world-wide pandemic.
At this point, we are over a year into this novel virus. We have run the gauntlet of personal protective equipment (PPE) shortages, food shortages, and toilet paper and paper towel shortages. Mask-wearing became political as cases exploded, and many fear there is no end in sight.
But maybe that’s no longer the case.
In October of 2020, Pfizer and Moderna became the first companies to announce that a vaccine designed to combat COVID-19 was close to completion. On December 11, 2020, the U.S. Food and Drug Administration (FDA) issued the first emergency use authorization (EUA) for the Pfizer vaccine to be used for the prevention of coronavirus disease 2019 (COVID-19) for individuals 16 years of age and older. On December 18, 2020, the FDA issued a second EUA for the Moderna vaccine to be used in the U.S. for those 18 years of age or older. With this exciting news, the end of the pandemic is in sight.
Maybe.
Many have felt skepticism that the vaccine was developed too quickly. How can a vaccine be designed in less than a year?
The mRNA vaccine—the design of both Pfizer and Moderna—has been in the works since 1989. In the early ‘90s, mRNA was advocated as a vaccine platform due to its strong safety advantages.1 An mRNA vaccine brings together the best of two worlds: the immunological features found in a live attenuated vaccine, such as endogenous antigen expression and T-cell induction, paired with those of killed or subunit vaccines, like defined composition and safety.1 Simply stated, while these vaccines are not live viruses, they invoke a strong T-cell response that provides us with ultimate protection.
The easiest way to understand how these vaccines can help bring about the end of this pandemic is to simply answer as many questions as possible, allowing everyone to make an educated decision about vaccination. According to the experts, such as Dr. Anthony Fauci and Dr. Peter Hotez, we need to achieve a population level of 80 percent vaccinated in order to acquire the herd immunity required to stop the transmission of the virus. The best way to achieve this is to educate the public.
How Does the mRNA Vaccine Work?
An mRNA vaccine has strands of genetic material—mRNA—inside a special coating. The coating protects the mRNA from enzymes in the body that would otherwise break it down. It also helps the mRNA enter the dendritic cells and macrophages in the lymph node cells near the vaccination site.
mRNA can be described as instructions for the cell on how to make a piece of the spike protein unique to SARS-CoV-2. Since only part of the protein is made, it does not harm the vaccinated person—but it is antigenic.
After the piece of spike protein is made, the cells break down the mRNA strand and dispose of it using enzymes in the cell. The mRNA never enters the cell’s nucleus or affects genetic material. Once displayed on the cell surface, the spike protein causes the immune system to begin producing antibodies, which means the immune system is primed to protect against future infection.
The action of the mRNA vaccine cannot alter a person’s DNA. As stated, the vaccine never enters the nucleus of the cell, and therefore cannot affect or interact with the DNA.2
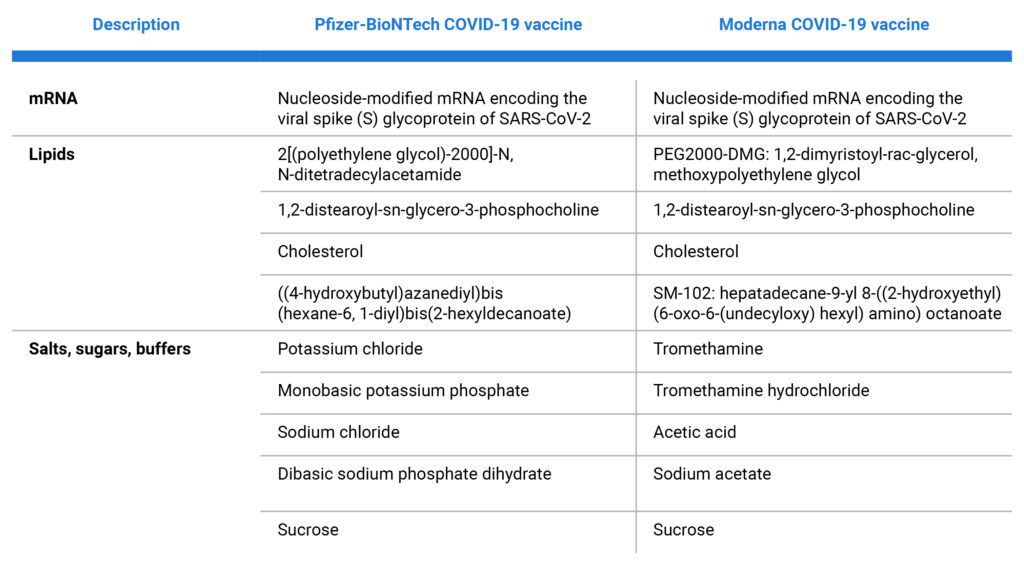
What Is in the Vaccines?
Refer to this chart for the components of the two available vaccines.3
Who Is Authorized to Receive These Vaccines?
Under the EAU, the following groups are authorized to receive the vaccine:
- Pfizer-BioNTech: ages 16 years and older
- Moderna: ages 18 years and older
How Should the Vaccine Be Administered?
- Pfizer-BioNTech: three weeks (21 days) apart
- Moderna: one month (28 days) apart
Persons should not be scheduled to receive the second dose earlier than recommended; however, second doses administered within a grace period of four days earlier than the recommended date are still considered valid. Doses inadvertently administered earlier than the grace period should not be repeated.
The second dose should be administered as close to the recommended interval as possible. If unable to adhere to the 21 or 28 days, the second dose of both Pfizer-BioNTech and Moderna may be administered up to six weeks (42 days) after the first dose.4
Are the Vaccines Interchangeable?
No. The mRNA COVID-19 vaccines are not interchangeable with each other or with other COVID-19 vaccine products. The safety and efficacy of a mixed product series have not been evaluated for safety at this time. Both doses of the series should be completed with the same product.4
What About Allergic Reactions?
You will be asked if you have ever had an allergic reaction to:
- Any component of a COVID-19 vaccine, including polyethylene glycol, which is found in some medications such as laxatives and preparations for colonoscopy procedures
- Polysorbate
- A previous COVID-19 vaccine
A history of any immediate allergic reaction to any other vaccine or injectable therapy is a precaution to currently authorized COVID-19 vaccines. While the vaccine may still be given, the patient should be counseled about unknown risks of developing a severe allergic reaction and balance these risks against the benefits of the vaccine.3
All recipients of the vaccine will be monitored for 15 minutes after the vaccine is given. Those with a history of allergic reactions will be monitored for 30 minutes.
Are Booster Doses Needed?
The need for and timing of booster doses for mRNA COVID-19 vaccines has not been established. Currently, no additional doses beyond the two-dose primary series are recommended.4
Should I Get the Vaccine if I Have Had or Currently Have COVID-19?
The simple answer is yes.
Vaccination of persons with an active case of SARS-CoV-2 infection should be deferred until the person has recovered from the acute illness and has met the criteria to discontinue isolation. Although there is some immunity following a case of SARS-CoV-2 infection, it is unknown how long that immunity lasts. Therefore, it is recommended that the vaccine be given for long-term immunity.4
For those who received treatment during their infection with monoclonal antibodies or convalescent plasma, vaccination should be deferred for 90 days after the initial infection to avoid potential interference of the antibody therapy with vaccine-induced symptoms.4
What if I Received Another Vaccine in the Last 14 Days?
The COVID-19 vaccine series should be administered alone, with a minimum of 14 days before or after administration of other vaccines.3 This is simply due to a lack of data on the safety and efficacy of mRNA COVID-19 vaccines administered simultaneously with other vaccines.
What if I Am Immunocompromised or Take Immunosuppressive Medications?
People with HIV infection or other immunocompromising conditions, or those who take immunosuppressive medications, might be at increased risk for severe COVID-19. The mRNA COVID-19 vaccines may be administered to people with underlying medical conditions who have no contraindications to the vaccination. Note, however, that there is the potential for reduced immune responses and the need to continue to follow all current protection guidelines, such as wearing a mask, social distancing, and washing hands frequently.3
What if I Am Pregnant or Breastfeeding?
If you are pregnant or breastfeeding, your healthcare provider may consider the level of COVID-19 community transmission, your personal risk of contracting COVID-19, the risks of COVID-19 to you and the potential risk to the fetus, the efficacy of the vaccine, the side effects of the vaccine, and the lack of data about use of the vaccine during pregnancy.3
Quite simply, pregnant women were not part of the study; therefore, always check with your provider before going ahead with the vaccine.
What Are the Vaccine’s Side Effects?
As stated earlier, mRNA vaccines are generally very safe and have minimal side effects. One way to think of the side effects is simply that your immune system is responding to the vaccine. If you experience them, your immune system is working to onboard the vaccine.
Immune responses seen with both the Pfizer-BioNTech and the Moderna vaccine are:3
- Fever, fatigue, and chills
- Pain, erythema, or swelling at the injection site
- Headache
- Vomiting or diarrhea may occur, but are rare
- Myalgia or arthralgia
It is important to note that there is a more pronounced immune response with the second vaccination. If you had the first vaccination and only had a sore arm, do not be surprised if you have all the above responses after the second. The good news is that they generally only last one to three days and, most importantly, it is your immune system working that sparks the response.
Is This the Beginning of the End?
On February 10, 2021, the CDC updated its guidance for those who have received both vaccinations. One of the reasons why we need to continue to wear our masks and social distance—even after vaccination—is because we do not know yet whether, following vaccination, the immune response will stop a COVID-19 infection immediately or if the person is a risk to those who have not been vaccinated. That very important question is still being studied; however, the guidance has changed for those who are fully vaccinated and have been exposed to someone with COVID-19.
Vaccinated people with an exposure to someone with suspected or confirmed COVID-19 are not required to quarantine if they meet all the following criteria:4
- They are fully vaccinated, meaning it has been two weeks since the receipt of the second dose of the vaccine.
- They have received the last dose of the series within the last three months.
- They have remained asymptomatic since the current COVID-19 exposure.
The simple fact that we can finally start to lighten up on any of the restrictions of the past year is an exciting step in itself!
Asking Questions Is How We’ll Get Through This
For all of you who, like me, want nothing more than to see the COVID-19 pandemic come to an end, education is our best defense. Ask questions, but only from those who know the facts. Do not rely on the news or social media for your answers. Talk to your healthcare provider or seek out the professionals on websites from organizations like the CDC or WHO. There are so many “theories” floating around about what this vaccine will do to us, when in reality it is one of the safest vaccines we have been given.
“Thousands of years ago, variola virus (smallpox virus) emerged and began causing illness and deaths in human populations, with smallpox outbreaks occurring from time to time. Thanks to the success of vaccination, the last natural outbreak of smallpox in the United States occurred in 1949. In 1980, the World Health Assembly declared smallpox eradicated (eliminated), and no cases of naturally occurring smallpox have happened since.”5
Why bring up smallpox? Because smallpox was successfully eradicated through a robust vaccination program. The last death due to smallpox was in 1978. The smallpox vaccine has a live virus in it, yet we received it, safely, knowing it was the right thing to do to protect ourselves from this devastating disease.
The latest predictions are that by September 2021, there will be enough of us vaccinated to return to a sense of normalcy. I am ready. How about you?
- Schlake, T., Thess, A., Fotin-Mleczek, M., & Kallen, K. J. (2012). Developing mRNA-vaccine technologies. RNA biology, 9(11), 1319–1330.
- CDC. (2021). Pfizer-BioNTech COVID-19 Vaccine Questions. Retrieved 2/14/2021 from https://www.cdc.gov/vaccines/covid-19/info-by-product/pfizer/pfizer-bioNTech-faqs.html
- CDC. (2021). Prevaccination Checklist for COVID-19 Vaccines. Retrieved 2/14/2021 from https://www.cdc.gov/vaccines/covid-19/downloads/pre-vaccination-screening-form.pdf
- CDC. (2021). Interim Clinical Considerations for Use of mRNA COVID-19 Vaccines Currently Authorized in the United States. Retrieved 2/14/2021 from https://www.cdc.gov/vaccines/covid-19/info-by-product/clinical-considerations.html
- CDC. (2021). Smallpox Home Page. Retrieved 2/14/2021 from https://www.cdc.gov/smallpox/